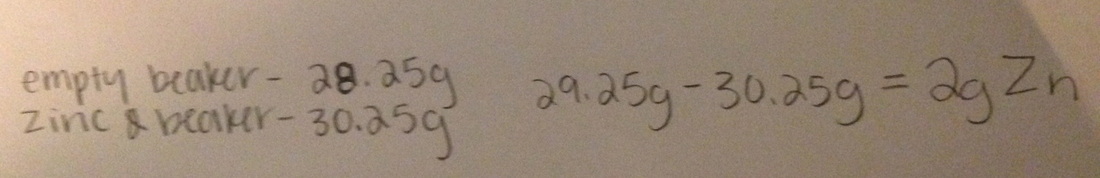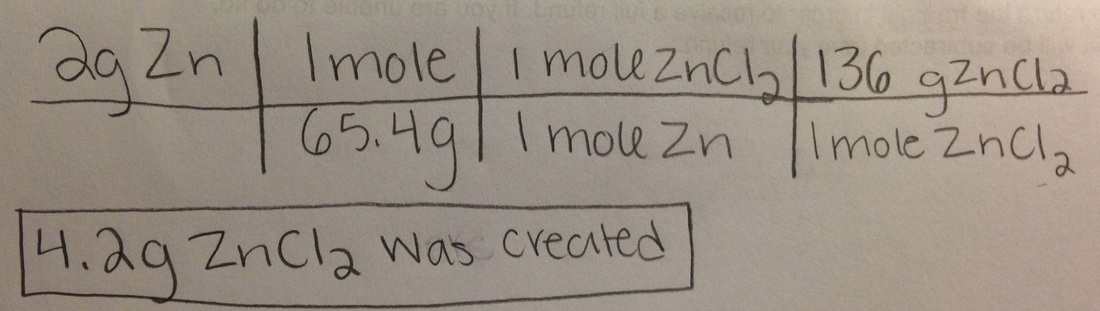Type of reaction: Single replacement
Reactants: cobalt (II) chloride and zinc
Product: ZnCl2
Reactants: cobalt (II) chloride and zinc
Product: ZnCl2
Balanced Reaction
CoCl2 + Zn → ZnCl2 + Co
Procedure
1. Since cobalt (II) chloride is a powder you must first dissolve it in water before a reaction can occur
2. After cobalt (II) chloride is fully dissolved in water drop the zinc in and observe the reaction
2. After cobalt (II) chloride is fully dissolved in water drop the zinc in and observe the reaction
Materials
-beaker
-water
-zinc
-cobalt (II) chloride
-stirrer
-water
-zinc
-cobalt (II) chloride
-stirrer